Currently, our researches focus on the interdisciplinary field of Nano, Materials Science, Chemistry, Catalysis and Energy. We are interested in the following directions:
1. Chemical hydrogen storage materials
Hydrogen is an ideal candidate as an energy carrier for mobile fuel cell applications, as it does not have any adverse effects on the environment, and is thus expected to be a promising energy vector for the near future. However, the problem of hydrogen storage is ofen considered to be a bottleneck for realization of the hydrogen economy. Ideally, a hydrogen storage system for mobile applications must possess a number of characteristics, including high storage capacity, fast kinetics, reversibility, cost efficiency, and safety under normal use. Among the many possible systems under research, a promising hydrogen storage technique based on liquid chemical hydrides with high hydrogen densities, in particular, cycloalkanes, nitrogen (and boron)-substituted heterocycles, methanol, formic acid, hydrous hydrazine, aqueous ammonia borane, and hydrazine borane, would meet the necessary requirements, while providing the additional advantage of good compatibility with the existing infrastructure for liquid fuels. Currently, we are searching new and efficient chemical hydrogen storage materials.
Hydrogen is an ideal candidate as an energy carrier for mobile fuel cell applications, as it does not have any adverse effects on the environment, and is thus expected to be a promising energy vector for the near future. However, the problem of hydrogen storage is ofen considered to be a bottleneck for realization of the hydrogen economy. Ideally, a hydrogen storage system for mobile applications must possess a number of characteristics, including high storage capacity, fast kinetics, reversibility, cost efficiency, and safety under normal use. Among the many possible systems under research, a promising hydrogen storage technique based on liquid chemical hydrides with high hydrogen densities, in particular, cycloalkanes, nitrogen (and boron)-substituted heterocycles, methanol, formic acid, hydrous hydrazine, aqueous ammonia borane, and hydrazine borane, would meet the necessary requirements, while providing the additional advantage of good compatibility with the existing infrastructure for liquid fuels. Currently, we are searching new and efficient chemical hydrogen storage materials.
References
1. Yadav, M. & Xu, Q. Liquid-phase chemical hydrogen storage materials. Energy Environ. Sci. 5, 9698-9725 (2012).
2. Zhu, Q.-L. & Xu, Q. Liquid organic and inorganic chemical hydrides for high-capacity hydrogen storage. Energy Environ. Sci. 8, 478-512 (2015).
3. Li, J., Zhu, Q.-L. & Xu, Q. Dehydrogenation of formic acid by heterogeneous catalysts. Chimia 69, 348-352 (2015).
4. Zhan, W., Zhu, Q.-L. & Xu, Q. Dehydrogenation of ammonia borane by metal nanoparticle catalysts. ACS Catal. 6, 6892-6905 (2016).
5. Yang, X. & Xu, Q. Gold-containing metal nanoparticles for catalytic hydrogen generation from liquid chemical hydrides. Chin. J. Catal. 37, 1594-1599 (2016).
6. Li, Z. & Xu, Q. Metal-nanoparticle-catalyzed hydrogen generation from formic acid. Acc. Chem. Res. 50, 1449-1458 (2017).
1. Yadav, M. & Xu, Q. Liquid-phase chemical hydrogen storage materials. Energy Environ. Sci. 5, 9698-9725 (2012).
2. Zhu, Q.-L. & Xu, Q. Liquid organic and inorganic chemical hydrides for high-capacity hydrogen storage. Energy Environ. Sci. 8, 478-512 (2015).
3. Li, J., Zhu, Q.-L. & Xu, Q. Dehydrogenation of formic acid by heterogeneous catalysts. Chimia 69, 348-352 (2015).
4. Zhan, W., Zhu, Q.-L. & Xu, Q. Dehydrogenation of ammonia borane by metal nanoparticle catalysts. ACS Catal. 6, 6892-6905 (2016).
5. Yang, X. & Xu, Q. Gold-containing metal nanoparticles for catalytic hydrogen generation from liquid chemical hydrides. Chin. J. Catal. 37, 1594-1599 (2016).
6. Li, Z. & Xu, Q. Metal-nanoparticle-catalyzed hydrogen generation from formic acid. Acc. Chem. Res. 50, 1449-1458 (2017).
2. Heterogeneous catalysis
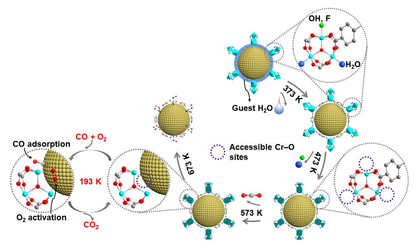
The synthesis of metal nanoparticles (MNPs) has drawn increasing attention in a broad range of catalytic processes, owing to their unique properties such as novel electrical structure, high fraction of surface atoms, and the quantum size effect. Currently, we are focusing on the synthesis of ultrafine and efficient MNPs and their applications including hydrogen evolution, CO oxidation, organic synthesis, and so on.
The synthesis of metal nanoparticles (MNPs) has drawn increasing attention in a broad range of catalytic processes, owing to their unique properties such as novel electrical structure, high fraction of surface atoms, and the quantum size effect. Currently, we are focusing on the synthesis of ultrafine and efficient MNPs and their applications including hydrogen evolution, CO oxidation, organic synthesis, and so on.
References
1. Jiang, H.-L. & Xu, Q. Recent progress in synergistic catalysis over heterometallic nanoparticles. J. Mater. Chem. 21, 13705-13725 (2011).
2. Zhu, Q.-L. & Xu, Q. Immobilization of ultrafine metal nanoparticles to high-surface-area materials and their catalytic applications. Chem 1, 220-245 (2016).
3. Yang, X., Sun, J.-K., Kitta, M., Pang, H. & Xu, Q. Encapsulating highly catalytically active metal nanoclusters inside porous organic cages. Nat. Catal. 1, 214-220 (2018).
4. Tsumori, N. et al. Quasi-MOF: exposing inorganic nodes to guest metal nanoparticles for drastically enhanced catalytic activity. Chem 4, 1-12 (2018).
1. Jiang, H.-L. & Xu, Q. Recent progress in synergistic catalysis over heterometallic nanoparticles. J. Mater. Chem. 21, 13705-13725 (2011).
2. Zhu, Q.-L. & Xu, Q. Immobilization of ultrafine metal nanoparticles to high-surface-area materials and their catalytic applications. Chem 1, 220-245 (2016).
3. Yang, X., Sun, J.-K., Kitta, M., Pang, H. & Xu, Q. Encapsulating highly catalytically active metal nanoclusters inside porous organic cages. Nat. Catal. 1, 214-220 (2018).
4. Tsumori, N. et al. Quasi-MOF: exposing inorganic nodes to guest metal nanoparticles for drastically enhanced catalytic activity. Chem 4, 1-12 (2018).
3. Multifunctional porous materials
3. Functional Metal-Organic Frameworks (MOFs).
a) for crystal designing.
b) as mesoporous materials.
c) as supports for nanocatalysts.
d) as storage materials for hydrogen and other gases.
4. Spectroscopic and Computational Studies on Reactions of metals and Small Molecules (CO, N2,...).
a) for crystal designing.
b) as mesoporous materials.
c) as supports for nanocatalysts.
d) as storage materials for hydrogen and other gases.
4. Spectroscopic and Computational Studies on Reactions of metals and Small Molecules (CO, N2,...).